The separation of arsenic metabolites in urine by high performance liquid chromatographyinductively coupled plasma-mass spectrometry
Article information
Abstract
Objectives
The purpose of this study was to determine a separation method for each arsenic metabolite in urine by using a high performance liquid chromatography (HPLC)- inductively coupled plasma-mass spectrometer (ICP-MS).
Methods
Separation of the arsenic metabolites was conducted in urine by using a polymeric anion-exchange (Hamilton PRP X-100, 4.6 mm×150 mm, 5 μm) column on Agilent Technologies 1260 Infinity LC system coupled to Agilent Technologies 7700 series ICP/MS equipment using argon as the plasma gas.
Results
All five important arsenic metabolites in urine were separated within 16 minutes in the order of arsenobetaine, arsenite, dimethylarsinate, monomethylarsonate and arsenate with detection limits ranging from 0.15 to 0.27 μg/L (40 μL injection). We used GEQUAS No. 52, the German external quality assessment scheme and standard reference material 2669, National Institute of Standard and Technology, to validate our analyses.
Conclusions
The method for separation of arsenic metabolites in urine was established by using HPLC-ICP-MS. This method contributes to the evaluation of arsenic exposure, health effect assessment and other bio-monitoring studies for arsenic exposure in South Korea.
Introduction
Arsenic, a metalloid and naturally occurring element, is one of the most abundant elements in the earth’s crust and is found throughout our environment. Arsenic is primarily used as an insecticide, as an herbicide due to its germicidal power, and as a preservative for wood to make it resistant to rot and decay resistant. Arsenic compounds are easily dissolved in water, therefore chronic arsenic effect from ingestion of arsenic-contaminated food and water or inhalation from the air has been investigated from many research groups in several countries. Arsenic exposure has been associated with detrimental health impacts such as hyperpigmentation, keratosis, skin and other types of cancer and vascular disease [1,2]. Because of the human health risk of arsenic, the International Agency for Research on Cancer (IARC) has classified arsenic as a group I human carcinogen [3]. It is believed that arsenic is a unique human carcinogen according to investigation into its carcinogenic effects over the last several decades. In regard to the carcinogenic effects of arsenic, recent studies have shown that cigarette smoking and ingestion of arsenic-containing drinking water at the same time significantly increased the synergistic effects of lung cancer [4]. In addition, other groups have suggested that a significant correlation between arsenic exposure and skin cancer has been observed [5]. There is also obvious evidence about the non-carcinogenic related effects of arsenic, including the chronic effect of arsenic exposure being strongly associated with spontaneous abortion and stillbirth [6], cardiovascular disease [7], disruption of hormonal systems [8], and type two diabetes mellitus [9].
Once arsenic compounds have been absorbed through various routes, such as the respiratory system or via food consumption, they are generally processed via a metabolic pathway in the liver [10], being converted into many different types of inorganic and organic species including arsenite (As3+), arsenate (As5+), dimethylarsinate (DMA), and monomethylarsonate (MMA). Arsenic metabolites in the body are eventually excreted into the urine, which is the major excretion pathway for the elimination of arsenic species from the body (Figure 1). Each arsenic form has a different physiological and bioactive property, hence it is necessary to identify and quantify each various arsenic chemical form for evaluation of human health risks related to arsenic exposure. In addition, previous studies have demonstrated that the total arsenic concentration, known as the sum of DMA, MMA and inorganic arsenic, is not sufficient for health risk assessment [11,12]. There have been several studies on the assessment of arsenic exposure in South Korea [13,14], but separation of arsenic metabolites in the general population in South Korea has not yet been reported. The toxicity of arsenic is strongly dependent on its chemical species present in the body. It is generally known that the inorganic forms of arsenic, such as As3+ and As5+, are more toxic than the organic arsenic, such as DMA and MMA, and among the inorganic arsenic species, As3+ is more toxic than As5+ [15-17]. In order to estimate human urinary arsenic metabolites, seafood ingestion should be prohibited at least 3 to 4 days prior to urine sample collection to avoid over estimation of urinary arsenic species. It is well known that some seafoods contain arsenic, including arsenobetaine in crustaceans and arsenosugars in seaweeds [18]. Thus, the aim of this study was to design an analytical procedure for separation of urinary arsenic metabolites by using high performance liquid chromatography (HPLC)-inductively coupled plasma-mass spectrometer (ICPMS). Using this method, it will contribute to the evaluation of arsenic exposure, human health effect assessment and other biomonitoring studies of arsenic exposure in South Korea.
Materials and Methods
Apparatus
For the separation of individual arsenic species, we used an Agilent Technologies 7700 series ICP-MS instrument (Agilent Technologies, Santa Clara, CA, USA), equipped with a lowflow sample introduction system and patented high matrix introduction kit. The HPLC system (Agilent Technologies) consisted of an Agilent 1260 Infinity Quaternary Pump VL, standard auto-sampler, and thermostat column compartment, equipped with Peltier cooling and heating apparatus for providing temperature stability and application flexibility. All samples were filtered with a 0.22 μm membrane before being placed into chromatographic vials (Agilent Technologies), then placed on the auto-sampler tray. A PRP X-100 anion-exchange HPLC column (5 μm, 4.6 mm×250 mm, Hamilton, Bonaduz, Switzerland) was used for arsenic species separation. The column was packed with a 55% cross-linked polystyrene divinylbenzene copolymer functionalized with quaternary ammonium anion-exchanger groups. The outlet of the column was directly connected to the sample introduction system of ICP-MS instrument. Reading data were collected by using Mass Hunter Workstation version B.01.01 (Agilent Technologies). The operating conditions for HPLC and ICP-MS are shown in Table 1.
Reagents
Deionized water was used for preparation and dilution of reagents, urine samples, and calibration solutions. Calibration solution supplied by Agilent Technologies, South Korea was prepared by dilution with deionized water from 1,000 mg/L stock. Standard reference material (SRM) and German external quality assessment scheme (G-EQUAS) No. 52 were used for internal control and accuracy of HPLC-ICP-MS. SRM 2669 was obtained from the National Institute of Standards and Technology (NIST) and G-EQUAS No. 52 was obtained from Erlangen- Nuremberg University, Germany. Prepared SRM was stored at -80°C until use. The reagents used in HPLC mobile phase, including ammonium carbonate, Tris, and ammonium sulfate (Sigma-Aldrich, St. Louis, MO, USA), were prepared in deionized water and filtered through a 0.45 μm membrane before use. These mobile phase reagents were prepared fresh daily. All reagents were analytical grade or better.
Linearity and Calibration Curves
Calibration was performed on five points over a concentration range of 1 to 20 μg/L. More than three calibration standards for each calibration point were prepared. Measurements for each calibration were averaged, then calibration curves were obtained, which resulted in good correlation coefficients of at least 0.9998.
Precision and Accuracy
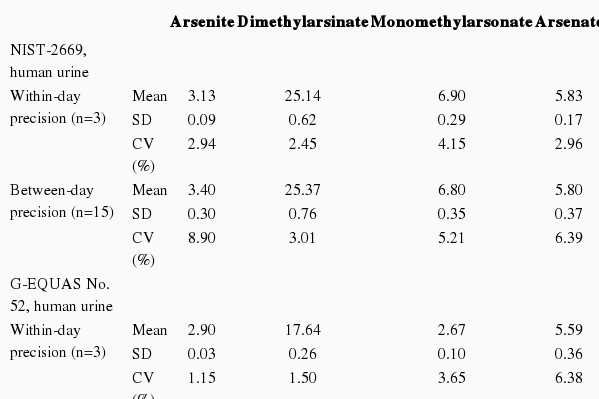
In order to validate the analytical procedure, within-day and between-day precision measurements were conducted by calculating the average, standard deviation (SD), and coefficient of variation (CV) using NIST SRM 2669 (Gaithersburg, MD, USA), and G-EQUAS No. 52. Accuracy was calculated by the recovery rate in all repeated analyses, using SRM 2669, and GEQUAS No. 52.
Statistical Analysis
For statistical analysis, SAS version 9.3 (SAS Institute Inc., Cary, NC, USA) was used. Data are expressed as the mean±SD of separate experiments.
Results
Linearity and Calibration Curves
To validate analysis accuracy, calibration was performed several times and the average signal intensity value for each calibration was used to make the calibration curves. The calibration curves of each five point arsenic species showed satisfactory linearity for As3+, As5+, DMA, and MMA (Figure S1). Retention times of arsenic metabolites, such as As3+, DMA, MMA, and As5+, are listed in Figure S2. The four major arsenic metabolites were separated within 16 minutes.
Repeatability, Accuracy, and Recovery rates
To validate repeatability and accuracy, we used two certified materials, NIST SRM 2669 and the G-EQUAS No. 52 urine sample. These certified materials provided the reference and tolerance values, enabling HPLC-ICP-MS to be used for the quantification of arsenic speciation. A reference values of four arsenic species in SRM 2669 were provided by NIST as follows 5.03 for As3+, 25.3 for DMA, 7.18 for MMA, and 6.16 for As5+, respectively. Reference values for G-EQUAS No. 52 were also provided by the manufacturer. Five replicate analyses with each certified material are summarized in Table 2, which show good results compared to the reference values. We further confirmed recovery rate for the separation of arsenic metabolites using two certified materials by HPLC-ICP-MS. Over 5 replicates samples of the certified materials were used for the recovery test. In the case of the G-EQUAS No. 52 samples, relative standard deviation values ranged from 8.38% for As3+, 4.12% for As5+, 3.44% for DMA, and 9.85% for MMA, respectively. Although recovery values gathered for all arsenic species were more diverse, values of over 90% were obtained through our arsenic species separation method. Even though other materials, including SRM 2669, provided small certified values, recovery values were almost the same as the G-EQUAS No. 52 sample, except for As3+ (Table 2). Within-day and between-day precision with two certified materials were also evaluated. CVs of four arsenic species in NIST SRM 2669 and G-EQUAS No. 52 sample revealed lower than 10% precision, respectively (Table 3). Those results clearly demonstrated that separation of arsenic metabolites in urine sample by using HPLC-ICP-MS could be an applicable method for the general population.
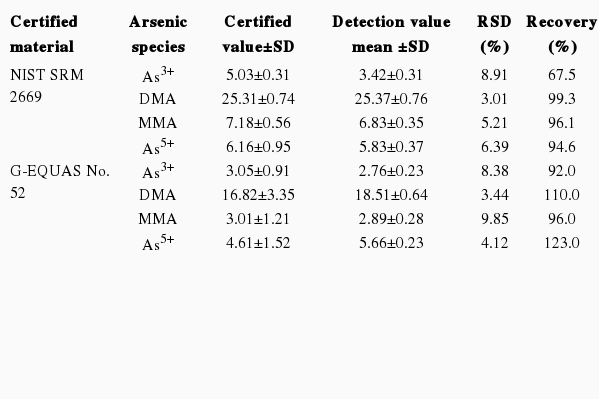
Recovery values of the arsenic metabolite separation analysis, using standard reference material (SRM) 2669 and G-EQUAS No. 52 samples, by HPLC-ICP-MS (μg/L)
Limit of Detection
Limit of detection (LOD) using HPLC-ICP-MS was also confirmed. To evaluate LOD, single standard solution (1 μg/L) was selected, then the concentration of the sample was determined over 10 times. The standard deviation for 10 samples was 0.05 for As3+, 0.09 for DMA, 0.09 for MMA, and 0.05 for As5+. The LOD of each arsenic species is summarized in Table 4.
Speciation of Arsenic in Urine Samples
To validate whether this method for separation of arsenic metabolites using HPLC-ICP-MS is appropriate for human urine samples, we further applied this method using five human urine samples. These samples were obtained from the Environmental Health Center, Dong-A University (IRB 13-063, Dong-A University Hospital). The concentrations of arsenic metabolites in these samples were obtained from five replicate HPLC-ICP-MS analyses, the results are shown in Table 5. Consistent with previous studies, results using human urine samples showed that DMA was the major arsenic metabolite in human urine samples. In addition, the sum of inorganic arsenic (As3+ and As5+) concentrations in the samples was shown to be within the range reported by the American Conference of Government Industrial Hygienists (ACGIH, 10 μg/L). In case of organic arsenic, since eating seafood was not strictly prohibited for 3 days before collecting urine samples, DMA results might have been affected by seafood ingestion.
Discussion
This study was performed to evaluate arsenic speciation in urine using a HPLC-ICP-MS technique. Arsenic is of interest in the field of arsenic metabolites separation analysis due to different toxicities being associated with different chemical forms. Inorganic arsenic, such as As3+ and As5+, is considered the most toxic form among arsenic metabolites, therefore arsenic is classified as genotoxic and carcinogenic by the IARC [19,20]. Previous studies have shown that chronic arsenic exposure can cause various human diseases. Recently, diabetes mellitus has been linked with arsenic-contaminated drinking water [21,22]. After arsenic consumption into the body, arsenic is metabolized into various types of arsenic chemical forms, then usually excreted in the urine, so urinary measurement is a major factor in biomarker determination for arsenic exposure assessment in humans. In this study, we have developed a method for separation of arsenic metabolites from urine using certified materials with HPLCICP- MS. All four major arsenic metabolites were separated within 16 minutes with detection limits ranging from 0.15 to 0.27 μg/L. It suggested that this method could be used for analyze the lower concentration of urinary arsenic metabolites present in human samples. A previous study conducted in 2010 using a HPLC-ICP-MS method showed detection limits ranging from 0.092 to 0.197 μg/L [23], which is a similar range to our detection limits, hence, this method for separation of arsenic metabolites could be considered as being a reliable analysis method for assessment of arsenic exposure.
Among arsenic metabolites, DMA is the main metabolite in urinary excretion under normal conditions without excessive ingestion of inorganic arsenic. Excretion ratios are usually 10%- 20% for inorganic arsenic, 10%-20% for MMA, and 60-80% DMA, respectively [24]. The present values for each arsenic species in our study using HPLC-ICP-MS show reasonable ratios in all arsenic metabolites and obtained results also show reliable ratios of each arsenic metabolite. The stability of retention time plays an important role in the reliable identification of each arsenic metabolite, because the identity of chromatographic peaks is normally obtained by comparing the retention time of the analysis of the sample with those of the standards. Therefore, we evaluated the retention time using a mixed calibration solution, then gathered recovery rates of each arsenic metabolite using certified materials. Using this method, calibration chromatograms were obtained from HPLC-ICP-MS analyses. Only 40 μL of sample had to be injected for the analysis, and excellent linearity measurements were obtained with r2=0.999. Further recovery rates using the G-EQUAS No. 52 certified material were in accordance with those obtained by others, whose recoveries were in the range of 87 to 105% for As3+, 82 to 112% for DMA, 89 to 111% for MMA, and 91 to 107% for As5+ [25,26]. Thus, we used this approach to check whether there was any interference with quantitative determination of arsenic species in urine samples. Analysis of five human urine samples using HPLC-ICP-MS revealed that inorganic arsenic species did not exceed the range reported by ACGIH. On the other hand, organic arsenic, especially DMA, showed higher results compared to other reports. Because, eating seafood was not strictly prohibited for 3 days before collecting these urine samples, it can be assumed that DMA concentrations might have been affected by seafood ingestion. It is well known that seafood ingestion can lead to misleading results, so seafood consumption is usually prohibited for at least 3 days before urine collection [18]. However, the current study introduces a method for separating arsenic metabolites in urine samples using HPLC-ICP-MS. We strongly suggest that continuous monitoring of the general population in Korea for arsenic metabolites in urine samples using HPLC-ICP-MS will provide significant information for the arsenic exposure index and contribute to the evaluation of health effect assessments from arsenic exposure in South Korea.
Acknowledgements
This research was conducted by the Heavy Metal Exposure Environmental Health Center at Dong-A University supported by the Ministry of Environment.
Notes
The authors have no conflicts of interest with the material presented in this paper.
References
SUPPLEMENTARY MATERIAL
Figure S1.
Calibration curves obtained from each arsenic metabolite (A) arsenite, (B) arsenate, (C) monomethylarsonate, and (D) dimethylarsinate, ranging from 1 to 20 μg/L using HPLC-ICP-MS. As3+, arsenite; As5+, arsenate; DMA, dimethylarsinate; MMA, monomethylarsonate.

Figure S2.
Chromatogram for arsenic metabolites obtained from HPLC-ICP-MS analysis. AB, arsenobetaine; As3+, arsenite; DMA, dimethylarsinate; MMA. monomethylarsonate; As5+, arsenate.